Abbott’s Alinity m Resp-4-Plex molecular assay, which was designed to test for four viruses in one test, has been authorized for emergency use by the US Food and Drug Administration (FDA). Shares of the global healthcare company gained 1.1% to close at $117.25 on March 5.
The Alinity m Resp-4-Plex test was introduced by Abbott (ABT) for detecting and differentiating SARS-CoV-2, influenza A, influenza B, and respiratory syncytial virus (RSV) in one test. This test will act as a rescuer as these four viruses have similar symptoms but need different treatments, the company said. Notably, the test is CE-marked and accessible in countries outside of the US.
Abbott Rapid and Molecular Diagnostics EVP Andrea Wainer said, “This newest test will allow for fast and efficient diagnosis and triage of patients who present with respiratory symptoms so they can be given the right care.”
The company has also received Emergency Use Authorization (EUA) for Alinity m SARS-CoV-2 test to detect individuals infected with SARS-CoV-2, but have no symptoms of COVID-19 infection. (See Abbott stock analysis on TipRanks)
On Jan. 28, BTIG analyst Marie Thibault upgraded the stock to Buy from Hold and maintained a price target of $140 (19.4% upside potential), following “a strong Q4 result, a blockbuster Diagnostics revenue result that beat consensus by more than $1B, and a bullish outlook for both 2021 and 2022 EPS.”
Given the company’s “faster growth rate for both sales and EPS, as well as its healthy balance sheet,” Thibault believes “ABT deserves to trade at a premium to the large-cap comp group.”
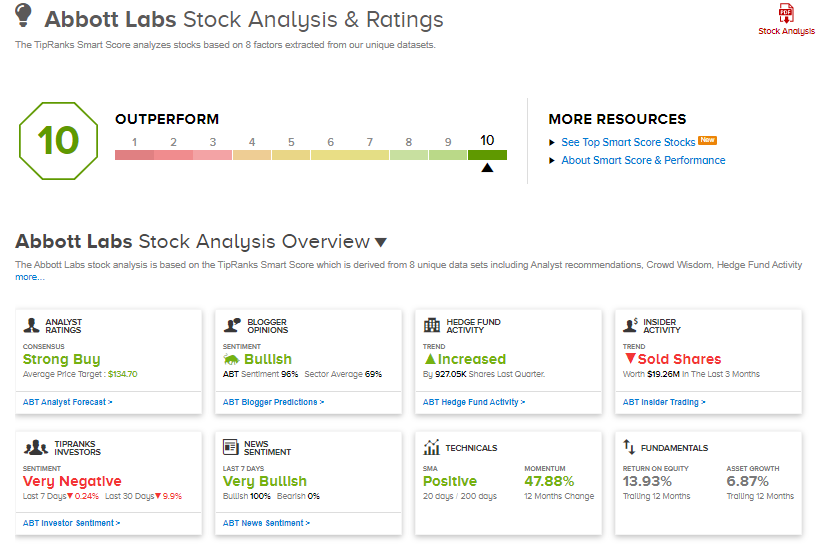
Abbott shares have exploded 52.2% over the past year, while the stock still scores a Strong Buy consensus rating based on 9 Buys versus 1 Hold. That’s alongside an average analyst price target of $134.70, which implies almost 15% upside potential to current levels.
Furthermore, Abbott scores a “Perfect 10” from TipRanks’ Smart Score rating system, indicating that the stock has strong potential to outperform market expectations.
Related News:
Amgen Inks $1.9B Deal To Buy Five Prime Therapeutics; Shares Pop 79%
Kroger Tops 4Q Profit Estimates As Online Sales Spike 118%; Shares Gain
BlueLinx Posts Blowout Quarter; Stock Rises 9%

